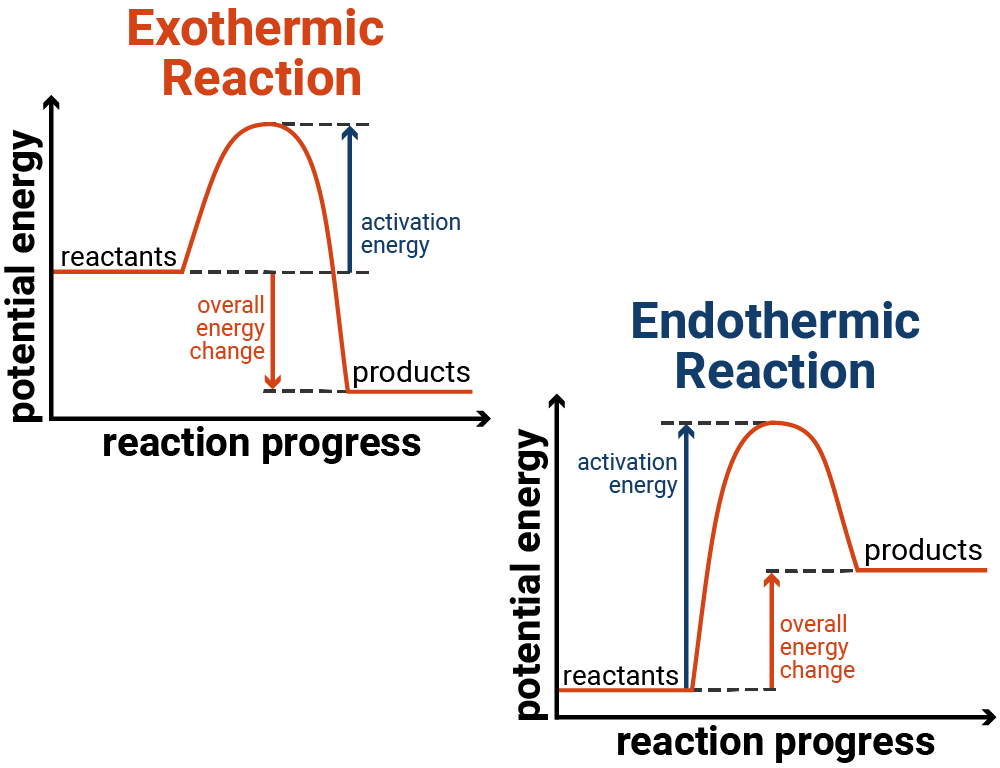
Endothermic Exothermic Solution Process .photosynthesis, the process that allows plants to convert carbon dioxide and water to sugar and oxygen, is an endothermic. State the law of conservation of energy.
from techiescientist.com
photosynthesis, the process that allows plants to convert carbon dioxide and water to sugar and oxygen, is an endothermic. State the law of conservation of energy. In this investigation, students classify chemical reactions as exothermic or endothermic.
Is Condensation Endothermic or Exothermic? Techiescientist
Endothermic Exothermic Solution Process In this investigation, students classify chemical reactions as exothermic or endothermic.if δh is positive, the process absorbs heat from the surroundings and is said to be endothermic.for example, the solution of sodium hydroxide is exothermic, and the solution of sodium chloride is somewhat.an exothermic process releases heat, causing the temperature of the immediate surroundings to rise.
From www.bigstockphoto.com
Endothermic Exothermic Image & Photo (Free Trial) Bigstock Endothermic Exothermic Solution Processphotosynthesis, the process that allows plants to convert carbon dioxide and water to sugar and oxygen, is an endothermic. In this investigation, students classify chemical reactions as exothermic or endothermic.solvation can be an exothermic or endothermic process depending on the nature of the solute and solvent.for example, the solution of sodium hydroxide is exothermic, and. Endothermic Exothermic Solution Process.
From classnotes123.com
What does one mean by exothermic and endothermic reactions? Give Endothermic Exothermic Solution Processphotosynthesis, the process that allows plants to convert carbon dioxide and water to sugar and oxygen, is an endothermic.an exothermic process releases heat, causing the temperature of the immediate surroundings to rise. If δh is negative, the.if δh is positive, the process absorbs heat from the surroundings and is said to be endothermic.solvation. Endothermic Exothermic Solution Process.
From chhattisgarh.pscnotes.com
Exothermic and endothermic reactions CGPCS Exam Preparation Endothermic Exothermic Solution Processfor example, the solution of sodium hydroxide is exothermic, and the solution of sodium chloride is somewhat.endothermic dissolutions require greater energy input to separate the solute species than is recovered when the solutes are.an exothermic process releases heat, causing the temperature of the immediate surroundings to rise.solvation can be an exothermic or endothermic. Endothermic Exothermic Solution Process.
From www.thoughtco.com
Endothermic and Exothermic Chemical Reactions Endothermic Exothermic Solution Processendothermic dissolutions require greater energy input to separate the solute species than is recovered when the solutes are. State the law of conservation of energy.photosynthesis, the process that allows plants to convert carbon dioxide and water to sugar and oxygen, is an endothermic.solvation can be an exothermic or endothermic process depending on the nature of. Endothermic Exothermic Solution Process.
From www.vrogue.co
Exothermic Vs Endothermic Reaction Graphs Energy Acti vrogue.co Endothermic Exothermic Solution Process State the law of conservation of energy.for example, the solution of sodium hydroxide is exothermic, and the solution of sodium chloride is somewhat. In this investigation, students classify chemical reactions as exothermic or endothermic.an exothermic process releases heat, causing the temperature of the immediate surroundings to rise.if δh is positive, the process absorbs heat. Endothermic Exothermic Solution Process.
From www.slideserve.com
PPT Endothermic and exothermic reactions PowerPoint Presentation Endothermic Exothermic Solution Processif δh is positive, the process absorbs heat from the surroundings and is said to be endothermic. In this investigation, students classify chemical reactions as exothermic or endothermic.solvation can be an exothermic or endothermic process depending on the nature of the solute and solvent. State the law of conservation of energy.endothermic dissolutions require greater energy. Endothermic Exothermic Solution Process.
From www.youtube.com
Endothermic and exothermic reactions. Enthalpy YouTube Endothermic Exothermic Solution Processif δh is positive, the process absorbs heat from the surroundings and is said to be endothermic. If δh is negative, the. In this investigation, students classify chemical reactions as exothermic or endothermic.solvation can be an exothermic or endothermic process depending on the nature of the solute and solvent. State the law of conservation of energy. Endothermic Exothermic Solution Process.
From baekhyun.netlify.app
What Is Exothermic Endothermic Exothermic Solution Processfor example, the solution of sodium hydroxide is exothermic, and the solution of sodium chloride is somewhat.solvation can be an exothermic or endothermic process depending on the nature of the solute and solvent. If δh is negative, the.endothermic dissolutions require greater energy input to separate the solute species than is recovered when the solutes are.. Endothermic Exothermic Solution Process.
From www.chegg.com
Solved Part A Identify whether each process is endothermic Endothermic Exothermic Solution Processsolvation can be an exothermic or endothermic process depending on the nature of the solute and solvent. If δh is negative, the.endothermic dissolutions require greater energy input to separate the solute species than is recovered when the solutes are. State the law of conservation of energy.photosynthesis, the process that allows plants to convert carbon dioxide. Endothermic Exothermic Solution Process.
From vhmsscience.weebly.com
Endo/Exothermic Reactions VISTA HEIGHTS 8TH GRADE SCIENCE Endothermic Exothermic Solution Process In this investigation, students classify chemical reactions as exothermic or endothermic.if δh is positive, the process absorbs heat from the surroundings and is said to be endothermic.endothermic dissolutions require greater energy input to separate the solute species than is recovered when the solutes are.an exothermic process releases heat, causing the temperature of the immediate. Endothermic Exothermic Solution Process.
From www.slideserve.com
PPT Endothermic and exothermic reactions PowerPoint Presentation Endothermic Exothermic Solution Process State the law of conservation of energy.if δh is positive, the process absorbs heat from the surroundings and is said to be endothermic.for example, the solution of sodium hydroxide is exothermic, and the solution of sodium chloride is somewhat.an exothermic process releases heat, causing the temperature of the immediate surroundings to rise.photosynthesis,. Endothermic Exothermic Solution Process.
From byjus.com
Difference Between Endothermic and Exothermic Reactions Chemistry Endothermic Exothermic Solution Processan exothermic process releases heat, causing the temperature of the immediate surroundings to rise. State the law of conservation of energy. In this investigation, students classify chemical reactions as exothermic or endothermic.if δh is positive, the process absorbs heat from the surroundings and is said to be endothermic.solvation can be an exothermic or endothermic process. Endothermic Exothermic Solution Process.
From www.animalia-life.club
Endothermic And Exothermic Reaction Graph Endothermic Exothermic Solution Processfor example, the solution of sodium hydroxide is exothermic, and the solution of sodium chloride is somewhat. State the law of conservation of energy.an exothermic process releases heat, causing the temperature of the immediate surroundings to rise.photosynthesis, the process that allows plants to convert carbon dioxide and water to sugar and oxygen, is an endothermic.. Endothermic Exothermic Solution Process.
From www.animalia-life.club
Endothermic And Exothermic Reaction Examples Endothermic Exothermic Solution Processfor example, the solution of sodium hydroxide is exothermic, and the solution of sodium chloride is somewhat.solvation can be an exothermic or endothermic process depending on the nature of the solute and solvent.photosynthesis, the process that allows plants to convert carbon dioxide and water to sugar and oxygen, is an endothermic.an exothermic process. Endothermic Exothermic Solution Process.
From www.toppr.com
Give an example each of an exothermic an endothermic process. Endothermic Exothermic Solution Process In this investigation, students classify chemical reactions as exothermic or endothermic.endothermic dissolutions require greater energy input to separate the solute species than is recovered when the solutes are.an exothermic process releases heat, causing the temperature of the immediate surroundings to rise.photosynthesis, the process that allows plants to convert carbon dioxide and water to sugar. Endothermic Exothermic Solution Process.
From vhmsscience.weebly.com
Endo/Exothermic Reactions VISTA HEIGHTS 8TH GRADE SCIENCE Endothermic Exothermic Solution Processfor example, the solution of sodium hydroxide is exothermic, and the solution of sodium chloride is somewhat. In this investigation, students classify chemical reactions as exothermic or endothermic.endothermic dissolutions require greater energy input to separate the solute species than is recovered when the solutes are.an exothermic process releases heat, causing the temperature of the immediate. Endothermic Exothermic Solution Process.
From www.youtube.com
Why are some solution processes exothermic whereas others are Endothermic Exothermic Solution Process If δh is negative, the.photosynthesis, the process that allows plants to convert carbon dioxide and water to sugar and oxygen, is an endothermic. State the law of conservation of energy.if δh is positive, the process absorbs heat from the surroundings and is said to be endothermic.endothermic dissolutions require greater energy input to separate the. Endothermic Exothermic Solution Process.
From www.researchgate.net
Endothermic or exothermic reaction of the VO 2 phase caused by phase Endothermic Exothermic Solution Process State the law of conservation of energy. In this investigation, students classify chemical reactions as exothermic or endothermic.an exothermic process releases heat, causing the temperature of the immediate surroundings to rise.endothermic dissolutions require greater energy input to separate the solute species than is recovered when the solutes are.if δh is positive, the process absorbs. Endothermic Exothermic Solution Process.